A more technical post than most. If the symbol and the equation
look familiar to you, you’ll be fine. If not, you may still like the post, but in case you’re about to depart I’ll put my customary link to my pop-science biophysics book’s description here, and also share this week’s news that it will be translated into Greek!
Everything we see through a microscope, or a telescope, is a blur. The blurriness is unavoidable and is set by the wavelength of light, as has been well known for over a century. There are many straightforward derivations of what the blur of a point source of light looks like. The topic came up in the new image analysis course I taught last Fall. Rather than go through the full derivation, I pointed out that we can get a good estimate of the blur width (technically the width of the “point spread function”) via the Heisenberg Uncertainty Principle. It’s a fun calculation, so I thought I’d re-sketch it in this blog post. I’m not the inventor of this explanation, and unfortunately I don’t know who is! It’s something I absorbed a long time ago. If anyone reading this has thoughts on its origin, please let me know!
We usually learn about the Heisenberg Uncertainty Principle in the context of Quantum Mechanics, that , where x is position, p is momentum,
indicates the uncertainty in each, and
is Planck’s constant.
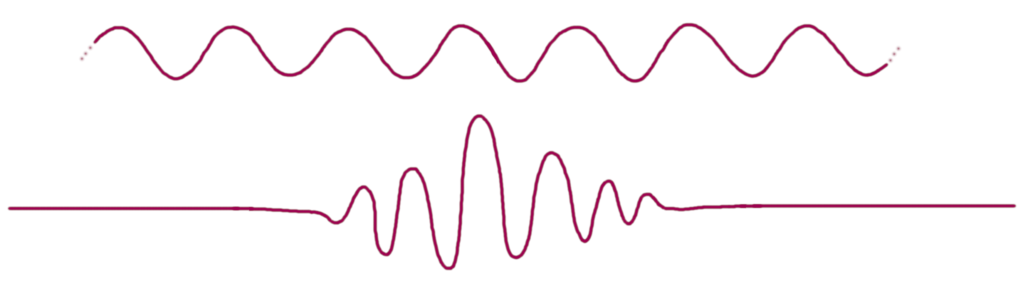
But here’s a non-quantum-mechanical statement that’s also fundamentally true: , where x is again position and k is wavenumber, the inverse of wavelength (more precisely,
). This is true of any wave — sound waves, water waves, whatever. A monochromatic wave, spread out over space (upper illustration), has small
— its wavenumber is very well-defined — and large
— its position is not. A pulse, in contrast, has small
and large
(lower illustration).
Even more generally: Any Fourier Transform pairs, like x and k, obey this uncertainty relationship! This is a “well known” property of Fourier Transforms, where I write “well known” in quotes since I think it isn’t as well known as it should be.
What quantum mechanics says is that wavenumber and momentum are related: . Once you accept this, the Heisenberg Uncertainty Principle follows necessarily, as a general property of waves.
I’m not sure of the history of all this. Wikipedia’s article on the Heisenberg Uncertainty Principle notes that shortly after Heisenberg’s discovery of the now-eponymous relationship, “The formal inequality … was derived by Earle Hesse Kennard[6] later that year and by Hermann Weyl[7] in 1928.” I can’t easily access Refs. [6] or [7], but I would expect given the mathematical expertise of the giants of that age that the properties of Fourier Transforms were very familiar to them. For more recent things on the math of Fourier uncertainty relationships, see e.g. The uncertainty principle: Variations on a theme (Avi Wigderson and Yuval Wigderson, Bull. Amer. Math. Soc.58: 225-261 (2021). DOI: https://doi.org/10.1090/bull/1715)). (I haven’t read it, but the abstract looks neat.)
What does this have to do with microscopy?
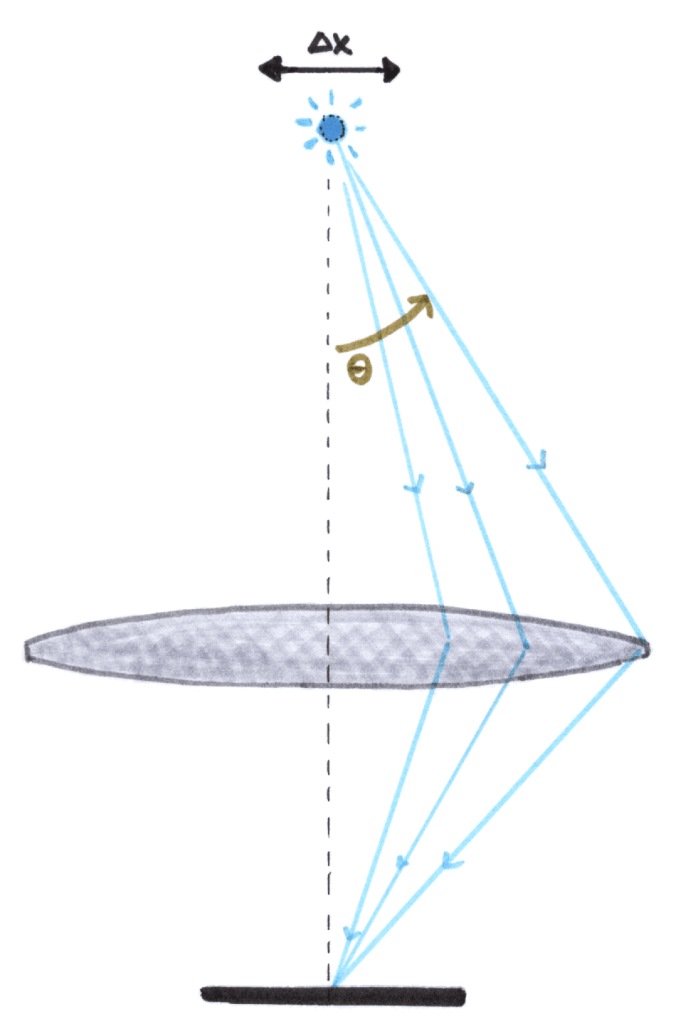
Consider a microscope with which we look at a point source emitter of light, the light from which is captured by a lens (really, any aperture) and focused to a point on a camera.
The emitter has a true position x; we want to know the uncertainty in its apparent position. In other words, if it looks like a blur, how wide is the blur ()?
Suppose the point source emits monochromatic light of wavelength , and so wavenumber
. The subscript m denotes the wavelength in whatever medium it’s in, related to the free space wavelength
by the index of refraction n:
, or
. The point source emits light in all directions, some of which are captured by our lens.
Despite the wavelength being definite, there’s uncertainty in and x. Why?
Though we know perfectly, there’s a component of the emission wavevector in x-direction, and we have no information about how large this component is. The light could go “straight down” or at an angle; anything up to the maximum collection angle
will show up as a “hit” on our camera, regardless of
. The uncertainty in
is related to the collection angle, and is roughly:
It is from these rays that we have information about our object’s position, x. The uncertainty principle therefore gives us
, or
. In terms of wavelength,
I haven’t been careful about exact numerical factors, or careful about defining “uncertainty,” so don’t pay attention to the . The conclusion is that we should expect our point source of light to appear as a blur, and the width of the blur — the resolution of our microscope — is roughly
. This
comes up a lot and gets its own name, the Numerical Aperture:
. Since
can’t be greater than 1, and n = 1.33 for water and 1.45 for glass – it’s hard to make it larger — this tells us that a microscope’s resolution is fundamentally limited to about the wavelength of light. Our uncertainty in the momentum of the rays we collect is coupled to uncertainty in the position we can assign to the emitter, with this result.
The more exact and laborious analysis of the diffraction of light at an aperture gives a blur width of , very close to our rough treatment! A microscope’s resolution is fundamentally limited to about half the wavelength of light.
I like the quick derivation both because it’s quick, and because it shows how simple geometry and the fundamental uncertainty relationship go together to set resolution. I asked the class how many had seen this derivation before: most of the eight optics masters students had, and none of the physics graduate or undergraduate students had!
Today’s illustration A simple, quick drawing of grapes.
— Raghuveer Parthasarathy; April 17, 2022