A terrible question to ask in science is unfortunately quite common: Does something affect something else? It seems innocuous enough — after all, one might want to know whether exposure to sunlight affects one’s risk of skin cancer, or whether the location of the moons of Jupiter affects the stock market. The answer, however, is always yes, and not realizing this is part of the plague of null hypothesis significance testing, p-values, and related ills. Everything affects everything else, and the correct questions to ask are how much? and Do I care about the magnitude of the effect? Skeptical? Here’s an example dating back over a century.
In 1914, the French mathematician and physicist Émile Borel noted that moving a gram of matter located far from our solar system by just one centimeter will sufficiently perturb the motion of gas molecules on Earth, due to the slightly shifted gravitational field, such that well within a second the molecules’ trajectories will be completely different than they would have been if the distant mass hadn’t moved. If we had perfect knowledge of all the positions and velocities of the gas molecules beforehand and could exactly predict their subsequent molecular motions, the motion of the distant matter would rapidly render our predictions useless. (Quantum mechanics makes this classical statement about predictability even worse.) Do little bits of stuff light years away affect gas molecules on Earth? Yes, and it doesn’t even take astronomical timescales, just fractions of a second. (Technically, it would take years for the gravitational perturbation to reach the Earth, which Borel was unaware of, but that’s irrelevant for the sub-second timescale of our gas being stirred up by the distant mass. Or put another way, the motion of stardust a few years ago suffices to make long-range prediction of molecular trajectories impossible.)
I’ll give a non-hypothetical example of astronomical motions detectably tugging on Earth-bound objects later, after tackling Borel.
In addition to highlighting the lack of isolation of any real system, Borel’s example points out the necessity and profundity of statistical mechanics. The details of nearly every system we care about, whether gas in a bottle, proteins in a cell, or electrons in a superconductor, are irrelevant and unknowable; it’s the statistical properties that matter. In fact, my undergraduate statistical mechanics course started off noting Borel’s work [1], and I’ve done the same in an undergraduate statistical mechanics course I’ve taught.
However, I’ve been annoyed for over a decade by how unclear Borel’s explanation is. Other writers have referred to it, perhaps most notably L. Brillouin in the 1960s [2], but as far as I’ve been able to find, they don’t actually derive Borel’s result. Every few years I think to search again, and I am always unsuccessful. I decided, therefore, to try to sketch a simple version of the calculation myself. You can feel free to skip to the numbers at the end, but in case you drop out completely, dear reader, I’ll include here my now customary brief advertisement of my upcoming pop-science biophysics book. You can read about it here or at the publisher’s site!
Reconstructing Borel’s Argument
Borel makes his statement in the book Le Hasard (1914). Not surprisingly, it’s in French. Despite watching a lot of old French crime movies, I have not picked up enough French to read Borel’s work, perhaps because the films involve long scenes of people silently digging their way out of prison rather than discussing anything. The miraculous Google Translate comes to my rescue, though, and twice — in 2007 and 2017 — I entered into it the relevant passages from Borel. He is verbose. An excerpt:
“Let us try to evaluate, from Newton’s law on universal attraction, the deviation which a molecule would undergo in the interval between two shocks, the displacement of an extremely small mass situated at a great distance… Let us now transport this miniscule sphere beyond the extremities of the visible universe, at a point from which the light takes billions of years to reach us…
… the effect of shock disperses very rapidly the bundles of trajectories supposedly infinitely untied, and the problem of the subsequent motion of the molecules becomes, of seconds, very indeterminate, in the sense that a colossally large number of different possibilities are a priori equally probable. … The only form in which the problem can be solved and solved is therefore the statistical form.”
(I posted PDFs of Borel’s text and Google’s translation; link below.) As others have also noted [3], Borel doesn’t place the moving mass at a distant star; this appears to be due to Leon Brillouin, decades later.
In another book, Introduction géométrique à quelques théories physiques (1914) Borel provides a longer argument that (at least in crude translation) is still not at all transparent, but that involves volumes in phase space and their transformation due to shifting forces — a sensible approach. I only stumbled on this work this year (2021). I’m again awed by the wonders of the internet and modern technology: the book is accessible on the Internet Archive (link) and Google Lens can be used to translate pieces from the images themselves. The relevant parts are in “NOTE II. Statistical Mechanics and Irreversibility,” pages 94-98.
Rather than slog through this, I made my own crude estimate of the effects of a distant shifted mass on the predictability of molecular trajectories. I suggest skipping the following paragraphs — the fun part is to see what approach you can think up yourself! How big is the change in trajectories induced by the changed gravitational field? What does it mean for the gas molecules trajectories to be “predictable” or not, and what is a criterion that distinguishes these two cases? See what you come up with…
The calculation
We imagine the usual sort of gas on Earth — what we might find in a balloon, for example — molecules of mass m, at temperature T and pressure P. The molecules zip around, and we can calculate their mean free path (the average distance between collisions) and the mean time between collisions
as usual [4]:
where T is temperature, P is pressure, and kB is Boltzmann’s constant.
Following Borel and Brillouin, we imagine M = 1 gram of matter far away; let’s say 4 light years (D = 4 x 1016 m), which moves away from us by = 1 cm.
This motion changes the magnitude of the gravitational force exerted on a local gas molecule by the distant mass, since its distance has increased. Specifically, the change in the force is
Now let’s make some leaps. I’m going to imagine perfect, classical, billiard-ball trajectories, and consider the system to have been rendered “unpredictable” if collisions between molecules that would have occurred do not occur, now that the distant mass has moved. This isn’t the only, or the best, way to think about predictability, but it’s simple and intuitive: if I can’t predict what molecules are going to collide, I can’t make (microscopic) predictions about the system. (Note that macroscopic behavior is still predictable!)
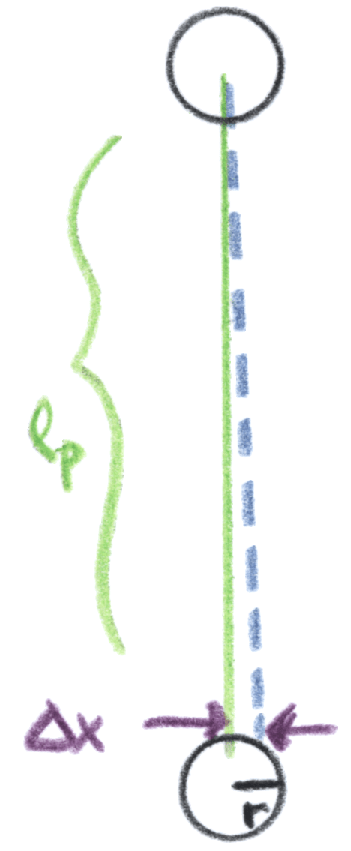
Imagine a gas molecule moving perpendicular to the vector pointing to the distant matter. Left alone, it would collide head-on with another molecule, let’s say. The change in force nudges it along that vector, perpendicular to its original path, so that by the time it collides it’s shifted by relative to its original position. How large is
, and will it be sufficient to avoid the collision entirely? You might object that this setup of perpendicular trajectories is the “best case” for a large effect. That’s true, but that’s ok — as we’ll see, we can relax this by diminishing the likelihood of an avoided collision by a factor of hundreds or thousands, with no change to the overall argument.
Ignoring factors of two, our , by elementary kinematics. Rather than hitting our (spherical” target molecule head-on, we hit at an angle
, where r is the radius of the molecule. Our trajectory upon reflection is therefore shifted by
from what it would be.
Now let’s think about the next collision. Even if we ignore the further gravitational effects of the distant mass, we get cascading effects from our initial shift. By the time it reaches the next molecule it collides with, our original molecule is displaced perpendicular to its original path by .
This causes a change in angle , which causes another displacement
, and so on. After n collisions, we have
.
Does this matter? What’s relevant is its magnitude compared to the radius of a molecule: if it’s much larger, the collision that would have happened will have been prevented. Let’s define a dimensionless factor
. Plugging in all the above expressions, including those for our mean free path and collision time,
If you’re following along, dear reader, now is a great time for dimensional analysis, making sure this is dimensionless. I’ve grouped together some dimensionless factors.
Even before plugging in numbers, we can see why our distant matter may perturb us: its effects multiply with every collision, scaling as a power of n. This will rapidly grow.
A problem with this argument. [Added later] My very clever colleague Eric Corwin points out a problem with this argument: “But shouldn’t you consider the differential change in force between one side of the sample and the other, instead? If everyone feels the same change in force then it just puts the whole system into a different accelerating frame and doesn’t do much about the internal collisions.” I had been imagining the effect of collisions by considering the “target” particle at rest relative to the moving one, a convenient setup for figuring out things like the mean free path, but Eric is correct that this won’t work here. Calculating the differential change in force across some distance would be fine, and I think everything would work similarly, perhaps extending the timescale of predictability, but I’m not certain. I’ve spent too long on this, so I will leave it as an exercise for the reader! (As far as I can tell, Borel doesn’t deal with this, either.) Update Nov. 29, 2021: I couldn’t stop myself from thinking about the differential force issue. The relevant “” gets an additional L / D factor, where L is the size of the system. The relevant size should be the mean free path,
, i.e. we care about a differential force across two colliding particles. The punchline is that all this has a negligible effect on our overall conclusions, as updated below in “numbers.” See the PDF linked immediately below for my sketch of the differential force calculation.
Now back to the original argument.
Now for some numbers
For molecular nitrogen, with a mass of m = 28 a.m.u. and a “kinetic radius” of 0.2 nm, and standard temperature and pressure, we find that by
! This corresponds to a time
, which is about 5 nanoseconds, given that the mean time between collisions for our gas molecules is about 1.6 ns. If we revisit our perhaps optimistic geometry and state that only 1 in 1000 collisions are perturbed by the amount we’ve calculated — a factor I’m recklessly making up, as being roughly the ratio of molecular radius to mean free path — our time to reach
increases by a factor of 1000, but it still very small: 5 microseconds. In other words, the 1-cm motion of a gram of matter 4 light years away makes prediction of molecular trajectories impossible beyond a timescale of a few microseconds.
Update Nov. 29, 2021: If I correctly account for the differential force across , as described above,
crosses 1 at about
rather than 30, or about 6 ns rather than 5 ns. With our fudge-factor of 1000, it’s still only 6 microseconds until unpredictability. The exponential scaling with n wins over any prefactor!
Imagine the effects of displaced materials at a much closer distance — the asteroid belt, or the room next door!
Can we detect it?
The answer to the question “Does X affect Y” is always, trivially, yes. This calculation is, perhaps, an entertaining example. Moreover, it’s yes with a guaranteed “p < 0.05” if one looks at enough samples. It’s perhaps worth keeping in mind concrete examples like the one above that highlight how essentially every effect size is nonzero, so that it’s clearer that the only meaningful questions are “how big is the effect?” and “how well can I estimate it?”
You may think this is example is silly since we’re not trying to calculate molecular trajectories, and in fact the lesson of all sorts of calculations like Borel’s is such a task is hopeless (even with computers far beyond any foreseeable capabilities) — we instead have to be clever, and realize that statistical properties are what matter, and these can be remarkably robust.
Nonetheless, it’s worth noting that these days we do observe the effects on Earth of the motion of distant objects. A stunning example is LIGO (the Laser Interferometer Gravitational-Wave Observatory), which uses interferometry to detect the tiny motion hanging mirrors causes by gravitational perturbations from far-off objects. How tiny? LIGO detects shifts in the mirror’s (average) position of about 10-18 meters , from stellar events hundreds of millions of light years away! [5] Watching a molecule be pushed a few nanometers by a few-light-year-distant chunk of rock doesn’t seem so implausible any more.
Today’s illustration.
I painted this based on a photo I took at a glacier in Iceland in September. It didn’t turn out as well as I had hoped, but it’s not as bad as I feared when I was halfway into it.
— Raghuveer Parthasarathy; November 28, 2021
NOTES and References
[1] That was Physics 112 at UC Berkeley, taught by the late Eugene Commins — an excellent course!
[2] e.g. L. Brillouin, Poincare’s theorem and uncertainty in classical mechanics. Information and Control. 5, 223–245 (1962).
[3] Bob Doyle, https://www.informationphilosopher.com/solutions/scientists/borel/
[4] See, for example, the first chapter of Schroeder’s very good textbook, “An Introduction to Thermal Physics”.
[5] On LIGO sensitivity, see e.g. the plot at https://ligo.org/science/Publication-S6CBCRangeDoc/index.php . On the distances of the events detected so far, see https://en.wikipedia.org/wiki/List_of_gravitational_wave_observations.




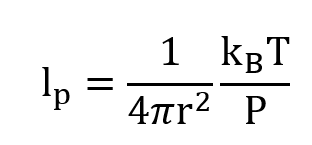
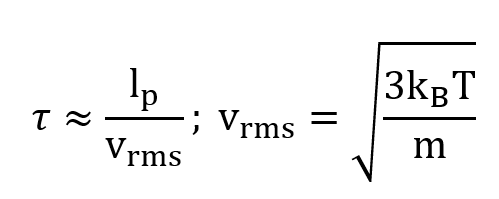
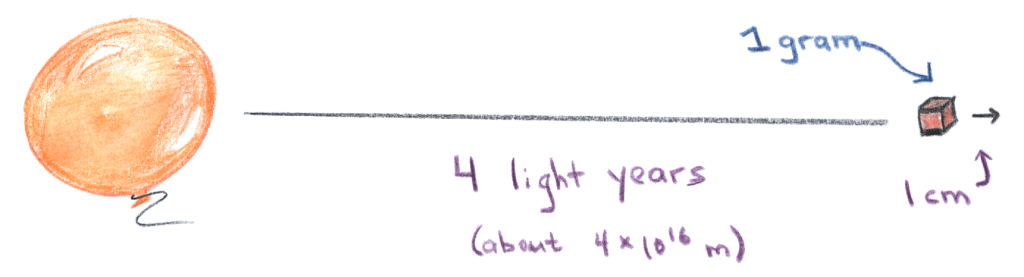
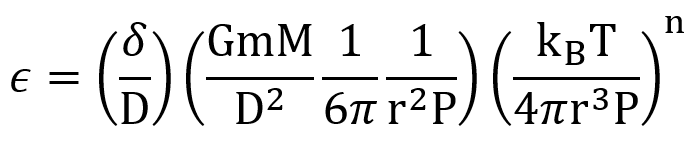
Dear Professor,
I doubt that a mass that is moved outside the solar system has any effect on anything on earth within a second, even on the path of molecules. Or does the gravitational pull manifest with superluminal speed?
Kurt Thomas
It’s a good question, but as I wrote: “Technically, it would take years for the gravitational perturbation to reach the Earth, which Borel was unaware of, but that’s irrelevant for the sub-second timescale of our gas being stirred up by the distant mass.” If the mass is 4 light years away, it means the motion 4 years ago (since gravity propagates at the speed of light) scrambles molecular trajectories now, and it makes them “unpredictable” on for timescales well within a second.
Fantastic post! Similar-but-different (right?) to the beloved “butterfly effect” in meteorology, i.e. because the atmosphere is a chaotic system, tiny differences in initial conditions rapidly grow, rendering accurate weather forecasts beyond roughly a ~2 week timescale impossible *in principle*, even if we had a perfect model.
Ed Lorenz 1963, http://journals.ametsoc.org/doi/abs/10.1175/1520-0469%281963%29020%3C0130%3ADNF%3E2.0.CO%3B2, gets the credit for this in atmospheric circles at least. Should Emile Borel get partial credit too?